systems biology
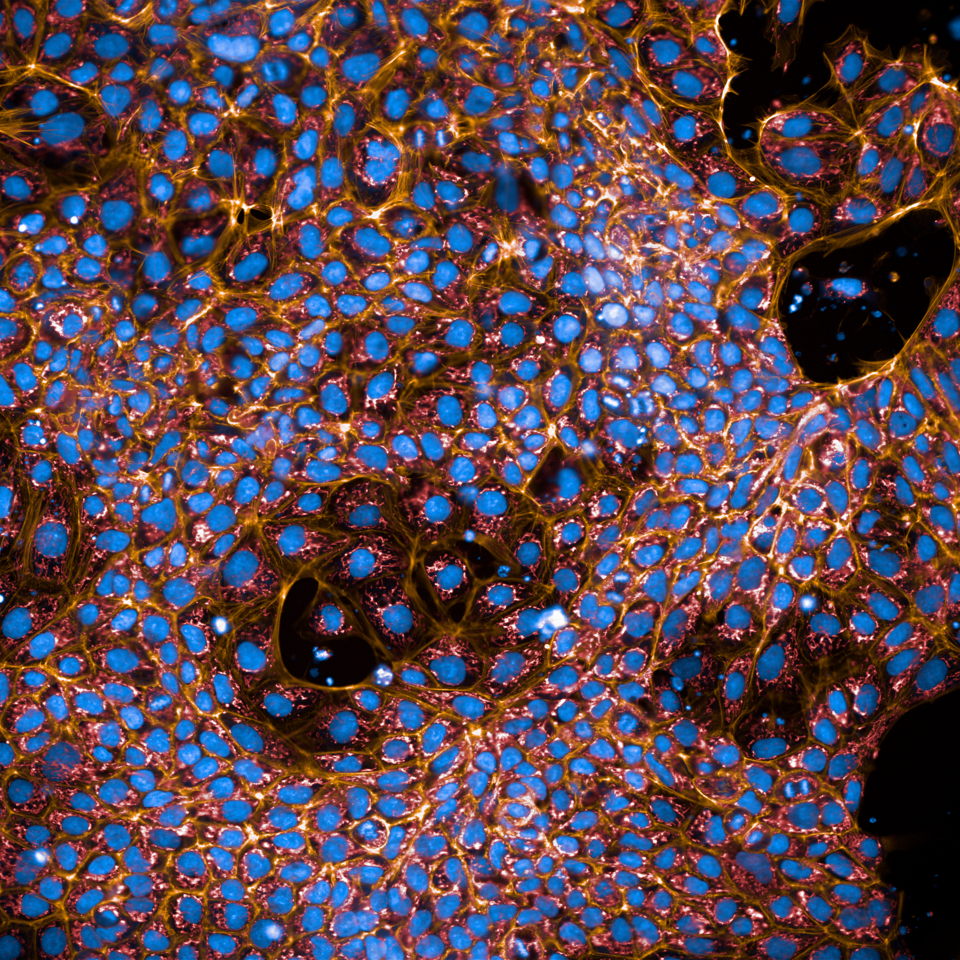
iPSCs stained with DAPI, Phalloidin (yellow) and CytC (red)
Image Courtesy of Snijder lab (Ramon Pfändler and Sohyon Lee)
__________________________________________________________
LABS |
|
_____________________________________________________________
|
|
Professor of Biology, Ernst Hadorn Chair Department of Molecular Life Sciences, University of Zurich Research Focus: We use in vitro grown populations of induced pluripotent stem cells to study cellular decision making by quantifying, at high throughput, large numbers of gene and protein activities simultaneously across thousands of single cells within the spatial context of the complex intracellular and multicellular environment. By covering multiple spatial scales relevant to cellular decision making within one sample, we can, for the first time, pinpoint the scale-crossing interactions that govern the collective behavior of molecules and cells and their responses to perturbations during cell differentiation. These insights will change the science of cells and cellular decision making and provide the missing logic of how individual cells can display activities and behaviors that are accurately tuned to the spatiotemporal context of a developing tissue. Methods: high-throughput automated microscopy, large-scale perturbations, subdiffraction imaging at scale, multiplexed protein state imaging (4i), multiplexed transcriptome imaging, computer vision, machine learning, data-driven modeling. Keywords: Genome-wide screening, image-based transcriptomics, crossing scales, computer vision, machine learing, ex vivo image-based diagnostics. Topics: Systems biology, single-cell biology, quantitative cell biology, phase separation, personalized medicine. Publications: https://pelkmanslab.org/publications/ Website: www.pelkmanslab.org _________________________________________________________________________top |
|
|
SNF Assistant Professor Institute of Molecular Systems Biology, ETH Zürich Research Focus: The Snijder Lab is interested in deciphering the molecular networks and organizational principles that drive cellular behavior in health and disease. To address these interests, we have pioneered multiplexed image-based drug screening in patient-specific biopsies, a technique we call pharmacoscopy, allowing us to measure patient-centered functional drug responses over hundreds of drugs at the single-cell level. Combined with multi-OMICs integration and deep learning-based image analysis, we aim to identify the molecular and cellular systems underlying human-to-human drug response variability and to improve patient treatment. In this context, we want to elucidate intra- and inter-donor variability in iPSCs derived from peripheral blood mononuclear cells (PBMCs) of a diverse cohort of healthy individuals. To address this, we aim to challenge donor-specific iPSCs in their pluripotent ground state or along differentiation paths, with the goal to reveal cell-intrinsic and cell-extrinsic drivers of phenotypic heterogeneity across individuals and to understand the molecular machineries underlying this variability. In particular, we are interested to unveil the contribution of metabolic states on the trajectory between naïve pluripotency and lineage restriction. Methods: high-content image-based functional screening (pharmacoscopy), PBMC reprogramming, Sendai virus, drug screening, transcriptomics, lipidomics Keywords: pharmacoscopy, high-throughput automated microscopy, multi-OMICs, human-to-human variability, iPSCs, heterogeneity, Topics: Systems biology, personalized medicine, single-cell biology, deep learning, molecular and cellular physiology Publications: https://www.snijderlab.org/#publications Webpage: https://www.snijderlab.org/ ___________________________________________________________________________top |

